Введение
В наступившем году исполняется 20 лет с момента выхода в свет первого отчета GOLD (Global Initiative for Chronic Obstructive Lung Disease), подготовленного сотрудниками Национального института сердца, легких и крови (США), а также международными экспертами, вошедшими в состав своего рода «мозгового центра», поддержанного Всемирной организацией здравоохранения [1]. Создание инициативы GOLD было продиктовано осознанием значительной и повсеместной распространенности хронической обструктивной болезни легких (ХОБЛ) и стремлением в связи с этим содействовать популяризации современных сведений о диагностике, лечении и профилактике заболевания. С тех пор инициатива GOLD заслуженно приобрела международное признание за разработку научно обоснованных стратегических документов, прежде всего ежегодных отчетов, освещающих ключевые вопросы ведения больных ХОБЛ. Знакомство с ними свидетельствует, в частности, о значительных инновациях, произошедших за 20-летний период в области разработки совершенных ингаляционных устройств, новых препаратов, знаний об их эффективных дозировках и потенциальных побочных эффектах, а также различных комбинаций лекарственных средств, используемых для облегчения симптомов, улучшения качества жизни и уменьшения числа повторных обострений заболевания.
При этом, несмотря на происходящие изменения в подходах к ведению больных ХОБЛ, одно оставалось неизменным, а именно признание за бронходилататорами ведущей роли в достижении фармакологического контроля заболевания [2], тем более что возможности этого направления терапии со временем претерпели радикальные изменения (рис. 1).
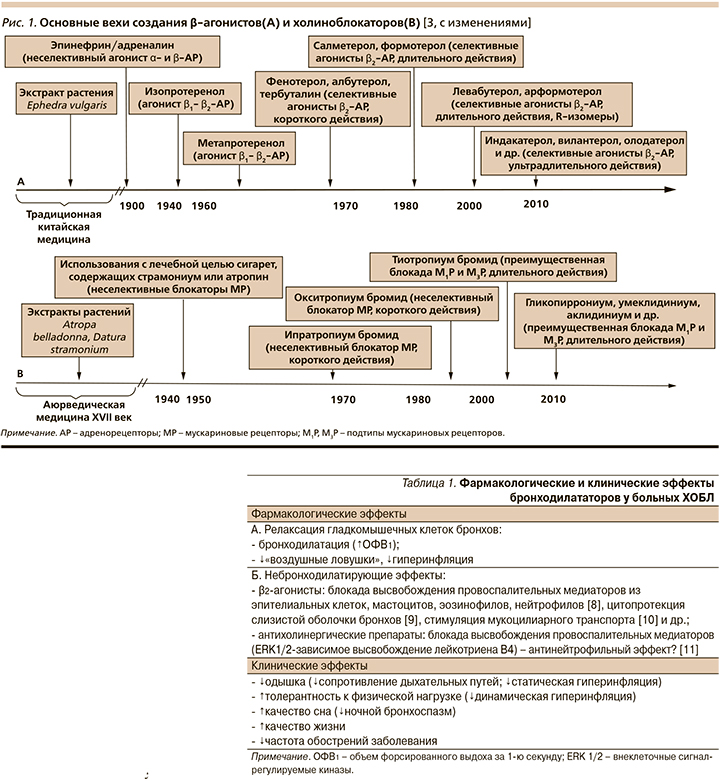
Место бронходилататоров в лечении ХОБЛ
Выбор бронходилататоров в качестве приоритетного направления лечения больных ХОБЛ аргументируется их многочисленными саногенетическими эффектами (табл. 1), достигаемыми главным образом прямым стимулирующим действием на β2-адренорецепторы (β2-агонисты) и непрямым – посредством блокады влияния ацетилхолина на мускариновые холинорецепторы (холиноблокаторы или антихолинергические препараты).
2010-е гг. ознаменовались появлением целого ряда длительно действующих β2-агонистов (ДДБА) и длительно действующих холиноблокаторов (ДДХБ) (табл. 2), а также их фиксированных комбинаций (табл. 3).
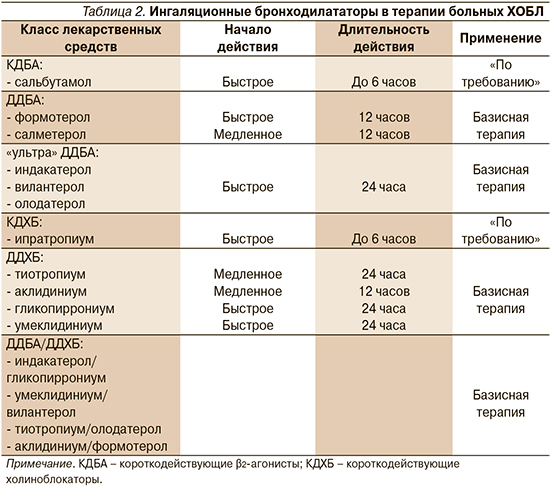

По мнению экспертов инициативы GOLD, фиксированные комбинации ДДБА/ДДХБ следует рассматривать как препараты «первой линии» для большинства пациентов с симптоматическим течением ХОБЛ и практически у каждого, кто нуждается в регулярной поддерживающей терапии [7]. Сходную точку зрения мы находим и на страницах большинства национальных рекомендаций по ведению больных ХОБЛ, в т.ч. и российских: назначение фиксированных комбинаций ДДБА/ДДХБ является приоритетным для всякого больного высокого риска независимо от клинического фенотипа, что определяется тяжелой/крайне тяжелой бронхиальной обструкцией или выраженной одышкой (mMRC1 ≥2) или по крайней мере двумя эпизодами нетяжелого обострения или потребовавшим госпитализации с одним эпизодом тяжелого обострения за истекший год или комбинаций указанных факторов [8, 9].
Фармакологическая основа кооперативного взаимодействия между β2-агонистами и холиноблокаторами очень сильна. Действительно, β2-агонисты вызывают бронходилатацию посредством релаксации гладкомышечных клеток бронхов независимо от характера констриктивных стимулов, т.е. выступают в качестве функциональных антагонистов бронхоконстрикции. В противоположность этому холиноблокаторы ингибируют бронхоконстрикторные эффекты ацетилхолина, вступая с ним в конкурентный антагонизм за взаимодействие с эффекторными мускариновыми рецепторами, дополняя и потенцируя функциональный антагонизм β2-агонистов [10].
К настоящему времени накоплен огромный фактический материал по оценке эффективности и безопасности «двойных» комбинаций ДДБА/ДДХБ, полученных в рамках плацебо-контролируемых исследований и исследований с активным контролем (ДДБА, ДДХБ и ингаляционные глюкокортикостероиды – ИГКС/ДДБА) [11, 12]. Так, согласно выводам недавно опубликованного систематического обзора и мета-анализа, включивших 23 исследования (20 185 больных ХОБЛ), фиксированные комбинации ДДБА/ДДХБ в ходе рандомизированных контролируемых исследований продолжительностью ≥12 недель превосходят монотерапию ДДХБ и ИГКС/ДДБА по величине прироста «наименьшего» (предбронхолитического) ОФВ1, причем в большинстве случаев этот прирост превышал минимальные клинически значимые величины (>100 мл).
Достоверным оказалось превосходство комбинированной бронходилатирующей терапии и во влиянии на выраженность одышки (транзиторный индекс одышки), формализованные критерии качества жизни (вопросник SGRQ2) и на частоту ситуационного применения бронходилататоров короткого действия [13].
При этом важно подчеркнуть, что назначение «двойных» бронходилататоров далеко не всегда следует рассматривать как этап эскалации терапии больного ХОБЛ, «не отвечающего» должным образом на монокомпонентное лечение. Хорошо известно, что значительная часть больных ХОБЛ не получают оптимального лечения [14, 15], тогда как раннее начало потенциально высокоэффективной терапии может обеспечить пациенту долгосрочные преимущества [16, 17]. Здесь же следует учитывать и тот факт, что начало лечения с более демонстративных в плане терапевтического эффекта препаратов способно склонить большее число больных ХОБЛ, традиционно характеризующихся низкой приверженностью [18], к более тщательному следованию врачебным рекомендациям.
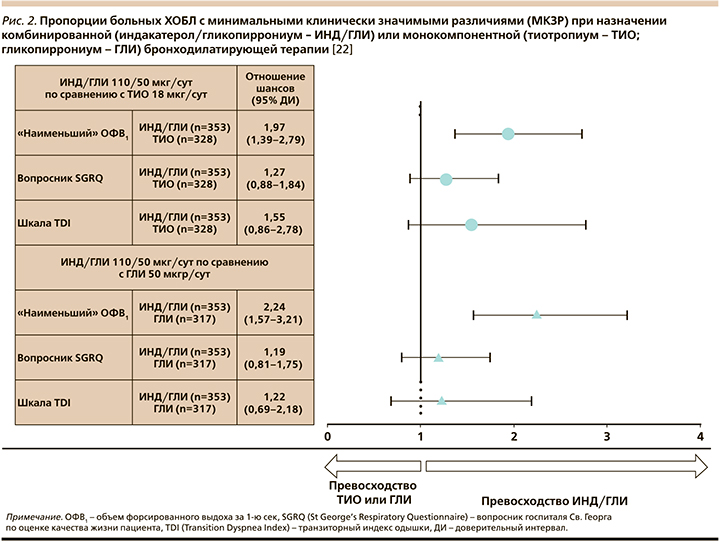
С учетом вышеизложенного становится все очевиднее обоснованность назначения «двойных» бронходилататоров в качестве терапии «первой линии» немалой части больных ХОБЛ. О пользе данной лечебной тактики свидетельствуют и результаты недавно проведенного объединенного анализа трех исследований – ARISE [19], SHINE [20] и SPARK [21], показавшего, что комбинация ИНД/ГЛИ превосходит монотерапию тиотропиумом и гликопиррониумом во влиянии на функцию легких, выраженность одышки и других симптомов ХОБЛ, качество жизни, частоту ситуационных ингаляций короткодействующих бронходилататоров у «наивных» больных, ранее не получавших регулярного лечения [22] (рис. 2).
Важнейшим с клинической точки зрения стало достоверное преимущество ДДБА/ДДХБ над ИГКС/ДДБА во влиянии на частоту среднетяжелых и тяжелых обострений ХОБЛ (отношение шансов [ОШ]=0,82; 95% доверительный интервал [ДИ]: 0,75–0,91) [13], ярко продемонстрированное при сравнении ИНД/ГЛИ и салметерола/флутиказона пропионата в исследовании FLAME [23] независимо от гендерных различий, возраста больных, статуса курения и степени тяжести заболевания [24].
Более привлекательным представлялся и профиль безопасности фиксированных комбинаций ДДБА/ДДХБ, по сравнению с ИГКС/ДДБА характеризовавшихся меньшей вероятностью развития побочных эффектов (ОШ=0,94; 95% ДИ: 0,89–0,99), в т.ч. и ятрогенной пневмонии (ОШ=0,59; 95% ДИ: 0,43–0,81), а также минимальной (по сравнению с ДДХБ) вероятностью прекращения терапии ввиду ее неэффективности (ОШ=0,66; 95% ДИ: 0,51–0,87) или (по сравнению с ИГКС/ДДБА) риском побочных эффектов (ОШ=0,83; 95% ДИ: 0,69–0,99) [13].
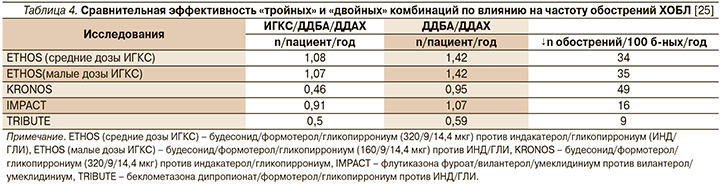
Вопрос о балансе пользы (прежде всего уменьшение частоты обострений ХОБЛ) и вреда (риск развития ятрогенной пневмонии) возник с особой остротой при появлении на фармацевтическом ранке «тройных» комбинаций (ИГКС/ДДБА/ДДХБ) и их сравнении с «двойными» бронходилататорами (табл. 4, 5).
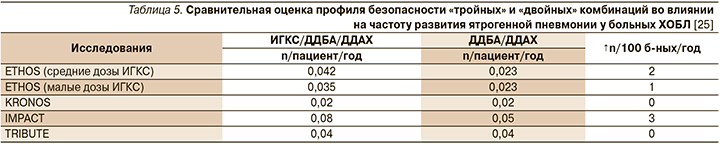
Имеющиеся на сегодняшний день данные позволяют утверждать, что применение «тройной» терапии не рассматривается в качестве стартового варианта лечения ХОБЛ и целесообразно только в подгруппе пациентов с более высоким исходным риском обострения заболевания (история обострения/обострений ≥1, потребовавших назначения системных антибиотиков и/или ГКС или обусловивших необходимость госпитализации в течение предыдущего года) [26]. Иными словами, ИГКС-содержащая терапия оправдана лишь в случаях повторных обострений ХОБЛ средней степени тяжести или единичных эпизодов тяжелого обострения, несмотря на продолжающийся прием ДДБА/ДДХБ, а также отдельных категорий больных, воспалительный профиль которых предполагает «ответ» на ИГКС (рис. 3) [27, 28].
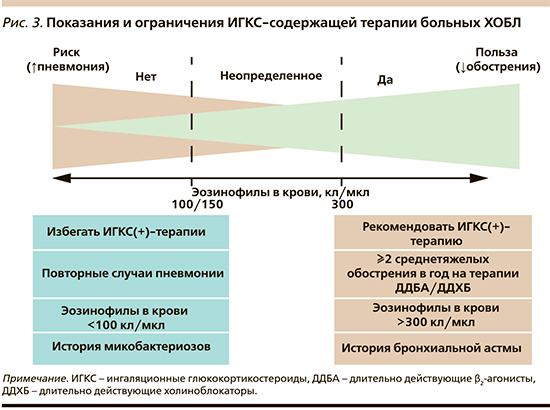
Данный подход разделяется и экспертами инициативы GOLD, ограничивающими показания к назначению ИГКС-содержащих комбинаций следующим перечнем клинических сценариев:
а) история госпитализаций в связи с обострением ХОБЛ у больного, получавшего длительно действующие бронходилататоры;
б) ≥2 среднетяжелых обострений ХОБЛ3 за год у больного, получавшего длительно действующие бронходилататоры;
в) эозинофилия крови > 300 /мкл (при проведении исследования вне обострения заболевания);
г) бронхиальная астма в качестве сопутствующего заболевания или имевшие место анамнестические указания на нее [7].
Как уже говорилось выше, в настоящее время доступны 5 фиксированных комбинаций ДДБА/ДДХБ, 4 из которых зарегистрированы в нашей стране (см. табл. 3). И в связи с этим возникает закономерный вопрос – являются ли продемонстрированные в ходе контролируемых исследований эффекты различных фиксированных комбинаций ДДБА/ДДХБ, в равной степени характерными для каждого из представителей этого класса лекарственных средств, а если нет, то какое клиническое значение могут иметь существующие между ними различия?
Однозначно ответить на этот вопрос сегодня не представляется возможным. Во-первых, до настоящего времени практически отсутствуют исследования прямого сравнения («head-to-head») отдельных представителей класса «двойных» бронходилататоров между собой. А те доступные единичные исследования [28] не свободны от ряда методологических ограничений (отсутствие плацебо-контроля, относительно небольшая продолжительность лечебного периода, открытый дизайн исследования и др.). Во-вторых, контролируемые исследования по оценке эффективности и безопасности каждого из «двойных» бронходилататоров отличались характеристиками пациентов, длительностью самих исследований, выбором препаратов активного контроля и «конечных точек» оценки эффективности.
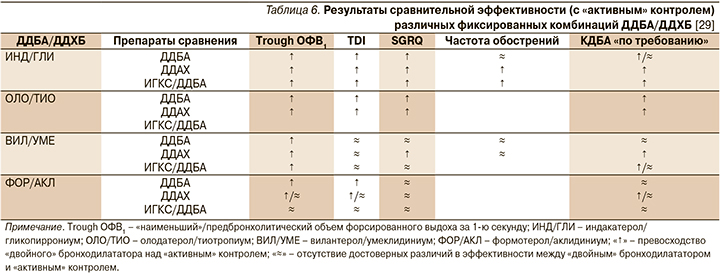
Тем не менее сравнение более или менее демонстративного «портфолио» каждого из обсуждаемых препаратов позволяет говорить об известном градиенте эффективности между ними [29] – табл. 6.
Потенциальная кардиотоксичность бронходилататоров для больных ХОБЛ
Обсуждение вопроса о роли и месте бронходилататоров в лечении ХОБЛ не было бы полным, если бы мы не затронули и такой важный аспект, как их потенциальная кардиотоксичность.
Хорошо известно, что стимуляция внелегочных β2-адренорецепторов при ингаляционном применении ДДБА может оказаться причиной неблагоприятных сердечно-сосудистых событий [30]. При этом установлено, что β1- и β2-адренорецепторы представлены в предсердиях и желудочках в примерных соотношениях 7:3 и 4:1 соответственно [31]. Присутствие β2-адренорецепторов, способствующих высвобождению норадреналина, обнаруживается и в адренергических нервных окончаниях в сердце [32]. Отсюда логичным выглядит предположение, будто, стимулируя β2-адренорецепторы, ДДБА могут вызывать положительные иноторпный и хронотропный эффекты, тем самым способствуя увеличению потребности миокарда в кислороде и развитию таких неблагоприятных сердечно-сосудистых событий, как тахикардия, эктопические нарушения ритма сердца, острая коронарная недостаточность [30, 33]. В дополнение к сказанному следует упомянуть и периферическую вазодилатацию вследствие стимуляции сосудистых β2-адренорецепторов, что может приводить к рефлекторной тахикардии [30].
Кроме того, ингаляционные β2-агонисты, оказывая стимулирующее воздействие на Na+, K+-АТФазу, связанную с β2-адоренорецепторами скелетных мышц, «перекачивают» ионы калия в клетку и тем самым могут вызывать гипокалиемию, которая в свою очередь рассматривается как фактор риска фибрилляций предсердий и желудочковой тахикардии [34].
Что же касается ДДХБ, то потенциальный сердечно-сосудистый риск при их применении связывают прежде всего с антагонизмом ко 2-му подтипу мускариновых (М2)-холинорецепторов синоатриального узла, что способно вызывать тахикардию [35]. Помимо этого в ряду механизмов «кардиотоксичности» ДДХБ указывается и ингибирование М3-холинорецепторов. Как известно, стимуляция М3-холинорецепторов защищает сердечную мышцу от ишемического повреждения путем активации антиапоптотических сигнальных веществ, повышения уровня эндогенных антиоксидантов, уменьшения внутриклеточной «перегрузки» Ca++ и др. [36, 37].
Однако, несмотря на указанные «предпосылки», результаты многочисленных контролируемых и наблюдательных исследований из реальной практики убеждают нас в обратном – длительно действующие бронходилататоры, в т.ч. и «двойные», не увеличивают риска развития серьезных сердечно-сосудистых событий по сравнению с активным контролем или плацебо [38].
Эта на первый взгляд противоречивая ситуация несоответствия «тревожных ожиданий» и оптимистичных выводов по результатам проведенных исследований может иметь несколько объяснений. Отчасти это связывается с тем, что больные ХОБЛ с сердечно-сосудистым риском часто исключаются из контролируемых исследований, обеспечивая результаты, отклоняющиеся от реальной жизни. Впрочем, в ряде исследований и post-hoc-анализов, дизайн которых предполагал включение пациентов с высоким риском кардиоваскулярных заболеваний, также не удалось выявить существенных различий в профиле сердечно-сосудистой безопасности между группами больных, получавших различную терапию, в т.ч. и «двойные» бронходилататоры [39, 40].
Более правдоподобным выглядит следующее объяснение – достигаемое при применении длительно действующих бронходилататоров и их «двойных» комбинаций улучшение дыхательной функции и уменьшение легочной гиперинфляции снижает и, вероятно, уравновешивает сердечно-сосудистые риски бронхолитической терапии больных ХОБЛ [41]. И для того чтобы аргументировать это предположение, следует обратиться к основным выводам исследования CLAIM, продемонстрировавшего улучшение сердечной деятельности на фоне 14-дневного приема ИНД/ГЛИ у больных ХОБЛ с умеренной/тяжелой гиперинфляцией [41]. Результаты этого исследования важны потому, что «двойная» бронходилатация, по-видимому, уменьшает известные вредные эффекты гиперинфляции на течение сопутствующих сердечно-сосудистых заболеваний у данной категории больных [38].
Таким образом, несмотря на бόльшую эффективность ДДБА/ДДХБ по влиянию на легочную функцию по сравнению с монокомпонентной терапией [42], «двойная» бронходилатация не сопровождается повышением риска развития неблагоприятных сердечно-сосудистых событий, а некоторые комбинации (в частности, ИНД/ГЛИ) могут играть даже своеобразную «кардиопротективную» роль [43, 44].
Ингаляторы у больных ХОБЛ
Очевидно, что ингаляционный путь введения является предпочтительным методом для доставки лекарственных аэрозолей в дыхательные пути [7], а эффективность ингаляционной терапии зависит в первую очередь от количества препарата, депонируемого в легких [45]. Однако здесь следует учитывать известную изменчивость дозы лекарственного средства, достигающего легких, что определяется рецептурой, типом устройства и способностью пациента осуществлять корректный ингаляционный маневр (табл. 7) [46–48].
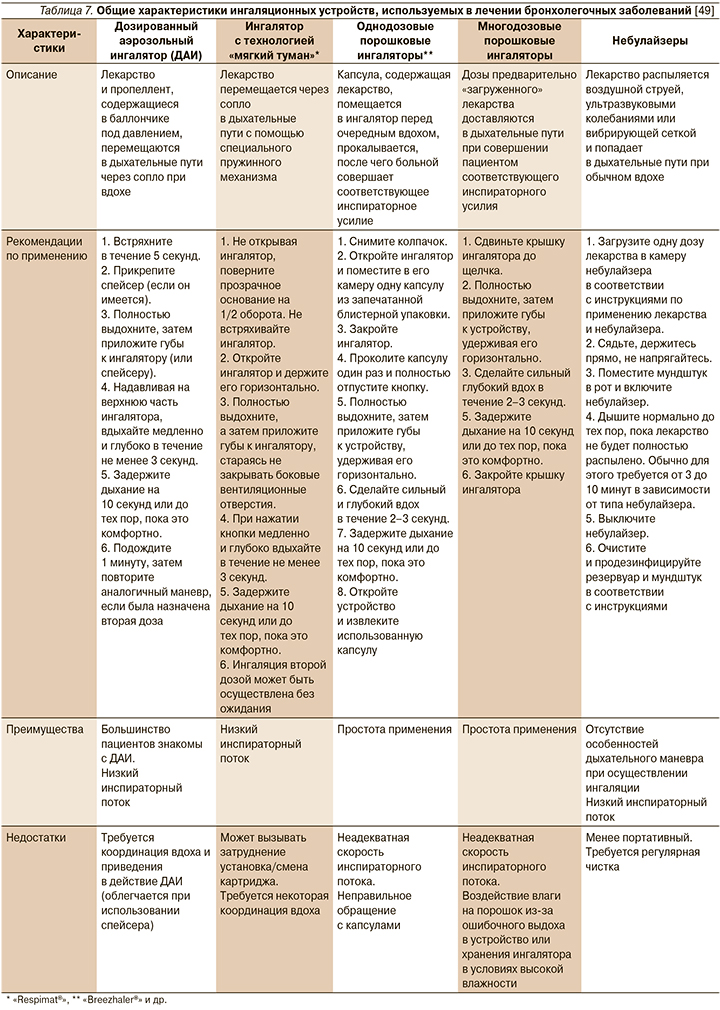
При выборе ингалятора необходимо принимать во внимание простоту и легкость при его использовании, что способствуют меньшему числу ошибок и обеспечивает более высокую приверженность длительной базисной терапии. Не менее важным является и такая характеристика ингаляционного устройства, как низкое сопротивление, что дает возможность осуществить вдох без особых усилий даже при выраженной бронхиальной обструкции [50].
При этом в случае использования дозированных порошковых ингаляторов с низким внутренним сопротивлением (например, «Breezhaler®») предполагает низкий максимальный перепад давления и не требует значительного усилия для создания разницы перепадов давления.
Мы формально должны рассчитывать на способность больного развить высокую скорость инспираторного потока [47]. А если при использовании ингалятора «Breezhaler®» пациент не будет способен развить скорость инспираторного потока ≥90 л/мин, справедливо задаться следующими вопросами: будет ли достигнута ожидаемая легочная депозиция лекарственного средства, препарат останется в капсуле, в ингаляторе, осядет на слизистой ротоглотки? На эти вопросы отвечает оригинальное исследование на платформе биофизической модели для изучения лекарственной депозиции с использованием Breezhaler® при различных скоростях инспираторного потока, углах наклона ингалятора и изменяемой анатомии дыхательных путей [51]. Авторами показано, что легочная депозиция ИНД/ГЛИ, 110/50 мг, индакатерола 150 мг и гликопиррониума 50 мг оказалась сопоставимой при различных скоростях (в диапазоне от 30 до 120 л/мин) инспираторного потока через Breezhaler® и не зависела от угла наклона ингалятора.
Эти данные совпадают с результатами ранее проведенного исследования, в ходе которого было продемонстрировано, что доставленные дозы индакатерола через Breezhaler® имеют минимальные отличия при скоростях инспираторного потоках от 30 до 100 л/мин [52].
К этому следует добавить, что при использовании ингалятора Breezhaler® пациентами допускается наименьшее число критических ошибок и они признают это устройство наиболее простым в использовании [53].
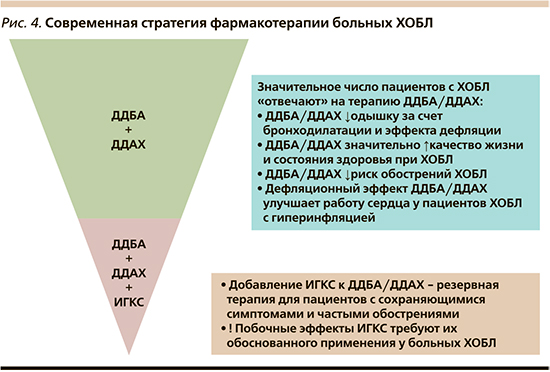
Заключение
Согласно инициативе GOLD, приоритетными целями в лечении больных ХОБЛ, проходящими «красной нитью» через все ее ежегодные отчеты, являются уменьшение риска повторных обострений и облегчение симптомов заболевания [1, 7]. И сегодня мы располагаем многочисленными свидетельствами того, что фиксированные комбинации ДДБА/ДДХБ, обеспечивая оптимальную бронходилатацию, играют первостепенную роль в достижении этих целей. Превосходя плацебо и активный контроль (ДДБА, ДДХБ, ИГКС/ДДБА), комбинированные бронходилататоры могут отличаться по своему терапевтическому потенциалу, хотя имеющаяся доказательная база (неравнозначная по числу, масштабу и дизайну исследований), в т.ч. и единичные прямые сравнительные исследования, не позволяют делать в каждом конкретном случае однозначный выбор в пользу той или иной фиксированной комбинации ДДБА/ДДХБ. Но в любом случае за «двойными» бронходилататорами прочно утвердился статус своего рода «краеугольного камня» в продолжающемся строиться здании эффективной и безопасной терапии ХОБЛ (рис. 4).


